-
1900: Walter Reed Yellow Fever StudyHuman Research
-
1904: First Federal Directive Stating Animals Should Not Be Subjected to Pain in Labs
Nonhuman Animal Research
This first federal directive, issued in 1904 by the Director of the Hygienic Laboratory, the first federal lab of medical bacteriology, marked a significant step in the ethical treatment of non-human animals in laboratory experiments. The directive emphasized that non-human animals should not be subjected to pain during research activities. It reflected a growing awareness and concern for non-human animal welfare within the scientific community, setting a precedent for the humane treatment of non-human animals in research settings.
-
1912: Public Health Service Established
Both
In 1912, under new authorizing legislation, the Marine Hospital Service was established as the Public Health Service (PHS) to express the enlarged scope of its work.
-
1930: Hygienic Laboratory Re-designated as NIH
Both
-
1932-1972: The US Public Health Service Tuskegee Study of Untreated Syphilis
in the Negro MaleHuman Research
-
1939: Stuttering StudyHuman Research
-
1946: Nazi Doctors' TrialHuman Research
-
1947: Nuremberg CodeHuman Research
-
1950: Animal Care Panel Established
Nonhuman Animal Research
In 1950, the Animal Care Panel was established to address growing concerns about the ethical treatment of non-human animals used in laboratory research. This initiative aimed to ensure that non-human animals received proper care and protection, reflecting a broader societal shift towards greater awareness of animal rights and welfare.
-
1950: NIH Issues Rules Regarding Animals
Nonhuman Animal Research
In 1950, NIH Director Dr. Rolla E. Dyer issues "Rules Regarding Animals," covering the ethical treatment and care of non-human animals in research.
-
1950: Radioactive Cereal Experiments at Fernald
Human Research
-
1953: First Federal Policy Introduced by NIH
Human Research
The first federal policy introduced by National Institutes of Health (NIH) (applied only to NIH intramural research) which, in addition to provisions for the protection of research participants, also required prospective review of research protocols. The inclusion of prospective review was consistent with the Nuremberg Code’s emphasis on the protection of healthy volunteers who had little to gain directly from research participation. Another important addition was the review of prospective research protocols by individuals who had no direct involvement with or intellectual investment in the research--marking the beginnings of the research review mechanism known today as Institutional Review Boards (IRBs).
-
1954: NIH Policy, Care, & Treatment of Lab Animals for PHS Funded Activities
Nonhuman Animal Research
The 1954 NIH Policy on the Care and Treatment of Laboratory Animals for Public Health Service (PHS) funded activities represented a landmark development in animal welfare regulations. This policy set forth guidelines for the humane care and treatment of non-human animals involved in research funded by the PHS, demonstrating a commitment to ethical standards in scientific endeavors.
-
1954: Rules Regarding Animals Revised as NIH Manual
Nonhuman Animal Research
In 1954, the Rules Regarding Animals were revised and consolidated into the NIH Manual, providing researchers with a comprehensive guide on the ethical and humane treatment of non-human animals in laboratory settings. This manual served as a valuable resource for ensuring the welfare of non-human animals used in biomedical research conducted under the auspices of the NIH.
-
1956: IAR Renamed ILARNonhuman Animal Research
The Institute of Laboratory Animal Resources (IAR) was renamed as the Institute for Laboratory Animal Research (ILAR) in 1956. This name change reflected a broader scope of activities and a growing emphasis on research related to laboratory animal science. ILAR continued to play a pivotal role in promoting the responsible and ethical use of non-human animals in research.
-
1956-1972: Willowbrook Experiment Human Research
-
1961-1962: Thalidomide StudyHuman Research
-
1963: Jewish Chronic Disease Hospital Study
Human Research
-
1963: Milgram StudyHuman Research
-
1963: Guide, 1st Edition
Nonhuman Animal Research
The first edition of "The Guide for the Care and Use of Laboratory Animals" was published in 1963 by the Institute of Laboratory Animal Resources (ILAR). This groundbreaking document set forth guidelines and recommendations for the proper care, housing, and humane treatment of non-human animals used in biomedical research. The Guide served as a foundation for the development of standardized practices and policies governing the use of laboratory non-human animals in scientific studies.
-
1964: Declaration of HelsinkiHuman Research
The Declaration of Helsinki was developed in 1964 by the World Medical
Association as an international statement of ethical principles to guide
medical professionals conducting research involving human subjects.
-
1965: AAALAC IncorporatedNonhuman Animal Research
The incorporation of the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) in 1965 marked a significant milestone in the establishment of accreditation standards for laboratory animal care programs. AAALAC's mission was to promote the humane treatment of non-human animals in research by accrediting institutions that adhered to high standards of animal care and welfare.
-
1966: Surgeon General's Directive: First Public Health Service (PHS) Policy
Human Research
Surgeon General's Directive: First Public Health Service (PHS) Policy -
required review of prospective research taking into account rights and
welfare of the subjects, appropriateness of the methods used to obtain
informed consent; risks and potential benefits of the research had to be
weighed; also instituted assurance requirement for grantee institutions.
-
1966: Public Law 89-544 Animal Welfare Act
Nonhuman Animal Research
Public Law 89-544, enacted in 1966, established the Animal Welfare Act (AWA) in the United States. This landmark legislation aimed to regulate the treatment of non-human animals in research, exhibition, transportation, and commerce, setting minimum standards for their care and housing. The AWA represented a major step forward in protecting the welfare of non-human animals used in various settings, including laboratories.
-
1966: Beecher Article Appears in the New England Journal of Medicine
Human Research
-
1967: ACP Renamed AALASNonhuman Animal Research
In 1967, the Animal Care Panel (ACP) underwent a name change and became known as the American Association for Laboratory Animal Science (AALAS). This renaming reflected the organization's evolving focus on promoting professionalism and best practices in the field of laboratory animal science, emphasizing the importance of animal welfare and ethical standards.
-
1971: Stanford Prison ExperimentHuman Research
-
1971: USDA Promulgated Subpart F (Stolen Animals)
Nonhuman Animal Research
In 1971, the United States Department of Agriculture (USDA) promulgated Subpart F of the Animal Welfare Act (AWA), addressing the issue of stolen non-human animals. This regulatory provision aimed to prevent the acquisition and use of stolen non-human animals in research, strengthening safeguards to ensure the ethical sourcing and treatment of non-human animals involved in scientific studies.
-
1971: NIH Policy, Care and Treatment of Laboratory Animals
Nonhuman Animal Research
The 1971 NIH Policy on the Care and Treatment of Laboratory Animals represented a significant advancement in promoting ethical standards for animal research funded by the National Institutes of Health (NIH). This policy outlined requirements for the humane care, handling, and treatment of laboratory non-human animals, emphasizing the responsibility of researchers to prioritize animal welfare in their work.
-
1972: Office for Protection from Research Risks Created
Human Research
The Office for Protection from Research Risks (OPRR), was created in 1972 as
part of the National Institutes of Health (NIH). (OPRR was later replaced by
OHRP).
-
1973: Expansion of NIH Policy to 1st PHS Policy
Nonhuman Animal Research
In 1973, the NIH Policy on the Care and Treatment of Laboratory Animals was expanded to become the first Public Health Service (PHS) Policy on laboratory animal care and treatment. This expansion broadened the scope of regulatory oversight to include all PHS-funded activities involving non-human animals, reinforcing the commitment to ethical standards and humane treatment in scientific research.
-
1974: National Research ActHuman Research
This directive, which was spearheaded by Senator Ted Kennedy and represented
one of his earliest achievements, created the National Commission for the
Protection of Human Subjects of Biomedical and Behavioral Research. The
Commission spent five years developing the Belmont Report and ultimately
mandated IRB review of all federally-funded research.
-
1974: OPRR EstablishedHuman
In 1974, the Office for Protection from Research Risks (OPRR) was established, signaling a commitment to safeguarding the welfare of human subjects involved in research funded by the Department of Health, Education, and Welfare (DHEW), which later became the Department of Health and Human Services (HHS). OPRR played a crucial role in developing policies and guidelines to ensure the ethical conduct of research and the protection of research participants.
-
1974-1981: Common Rule and FDA Regulations
Human Research
-
1976: FDA Proposed GLPNonhuman Animal Research
The U.S. Food and Drug Administration (FDA) proposed Good Laboratory Practice (GLP) regulations in 1976, aiming to ensure the quality and integrity of non-clinical laboratory studies conducted for the evaluation of pharmaceuticals, food additives, and medical devices. These proposed regulations outlined standards for the conduct, documentation, and reporting of studies to support the safety and efficacy of regulated products, enhancing the reliability of preclinical research.
-
1977: FDA Guidance Issued - General Considerations for the Clinical Evaluation
of DrugsHuman Research
-
1978: Guide Revision 5th Edition
Nonhuman Animal Research
The publication of the 5th edition of the Guide for the Care and Use of Laboratory Animals in 1978 represented a significant update to the guidelines established by the Institute for Laboratory Animal Research (ILAR) and the National Research Council (NRC). This revised edition provided updated recommendations for the housing, care, and use of laboratory non-human animals, reflecting advancements in scientific knowledge and evolving standards of animal welfare.
-
1979: Belmont ReportHuman Research
-
1979: FDA GLP Became LawNonhuman Animal Research
In 1979, the U.S. Food and Drug Administration's (FDA) Good Laboratory Practice (GLP) regulations became law, establishing requirements for the conduct of non-clinical laboratory studies supporting the safety and efficacy of FDA-regulated products. This regulatory milestone standardized procedures for study conduct, data documentation, and quality assurance, ensuring the reliability and integrity of research submitted to the FDA for product approval.
-
1979: USDA Promulgated Subpart E (ID of Animals)
Nonhuman Animal Research
The United States Department of Agriculture (USDA) promulgated Subpart E of the Animal Welfare Act (AWA) in 1979, focusing on the identification of non-human animals used in research settings. This regulatory provision established requirements for the proper identification of non-human animals involved in covered activities, facilitating traceability and accountability in research involving animal subjects.
-
1979: PHS Animal Welfare Policy Revised, PHS Assurance Required
Nonhuman Animal Research
The revision of the Public Health Service's (PHS) Animal Welfare Policy and
the requirement for PHS Assurance emphasized the commitment to the humane
care and use of non-human animals in research funded by the PHS. This
development highlighted the importance of ethical oversight and responsible
conduct in research involving non-human animals, promoting the well-being of
research subjects and the integrity of scientific inquiry.
-
1983: IRACNonhuman Animal Research
The Interagency Research Animal Committee (IRAC) was formed in 1983 to address issues related to the care and use of laboratory non-human animals across federal agencies involved in research. Comprising representatives from various agencies, IRAC aimed to harmonize policies, share expertise, and coordinate efforts to enhance the welfare of non-human animals used in research while ensuring scientific rigor and regulatory compliance.
-
1984: FDA Proposed Amendments to GLP Regs
Nonhuman Animal Research
In 1984, the Food and Drug Administration (FDA) proposed amendments to the Good Laboratory Practice (GLP) regulations. These proposed changes aimed to enhance the quality and integrity of non-clinical laboratory studies conducted for regulatory submissions, including those involving animal testing.
-
1984: US Government Principles Promulgated
Nonhuman Animal Research
In 1984, the US Government Principles Regarding the Care and Use of Animals in Testing, Research, and Training were promulgated. These principles provided overarching guidance for the ethical treatment of non-human animals involved in research, emphasizing the importance of minimizing pain and distress, ensuring appropriate housing and care, and considering alternatives to animal use whenever possible.
-
1985: AWA Amendments Passed By Congress
Nonhuman Animal Research
In 1985, amendments to the Animal Welfare Act (AWA) were passed by Congress, requiring all registered research institutions to establish Institutional Animal Care and Use Committees (IACUCs) to oversee non-human animal research and approve proposed protocols. The revised Public Health Service (PHS) policy on non-human animal research also mandated the creation of IACUCs for institutions receiving PHS funds. These amendments emphasized the importance of minimizing pain and distress in non-human animal subjects and encouraged the exploration of alternatives to potentially painful research procedures.
-
1985: HREA Passed by CongressNon-human
The Humane Research and Experimentation Act (HREA) was passed by Congress in 1985, introducing further regulations to promote the ethical treatment of non-human animals in research. This legislation aimed to establish clear standards for the care and use of non-human animals in scientific studies, emphasizing the importance of minimizing pain and distress, providing appropriate housing and veterinary care, and considering alternatives to animal experimentation whenever feasible.
-
1985: Guide Revision 6th Edition
Nonhuman Animal Research
In 1985, the sixth edition of the Guide for the Care and Use of Laboratory Animals was revised. This widely recognized guide provided comprehensive recommendations for the proper care, housing, and use of non-human animals in research, education, and testing.
-
1986: AWIC EstablishedNonhuman Animal Research
In 1986, the Animal Welfare Information Center (AWIC) was established to facilitate access to information related to the humane care and use of non-human animals in research, testing, and teaching. Operating within the National Agricultural Library of the United States Department of Agriculture (USDA), AWIC serves as a valuable resource for researchers, educators, and regulatory agencies seeking guidance on animal welfare issues.
-
1986: PHS Policy Revised to Include HREA
Nonhuman Animal Research
In 1986, the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals was revised to incorporate provisions of the Humane Research and Experimentation Act (HREA). This revision aimed to align PHS policy with legislative requirements, ensuring that institutions receiving PHS funding adhered to established standards for the ethical treatment of non-human animals in research.
-
1987: FDA Final Rule Amending GLP Regs
Nonhuman Animal Research
In 1987, the Food and Drug Administration (FDA) issued a final rule amending the Good Laboratory Practice (GLP) regulations. These amendments represented the culmination of regulatory efforts to enhance the quality and reliability of non-clinical laboratory studies conducted for regulatory purposes.
-
1986: Report of AVMA Panel on Euthanasia
Nonhuman Animal Research
In 1986, the American Veterinary Medical Association (AVMA) released a report from its Panel on Euthanasia, addressing best practices for the humane euthanasia of non-human animals in various settings, including research laboratories. The report provided guidance on appropriate methods and techniques for euthanasia, emphasizing the importance of minimizing pain and distress while achieving a rapid and peaceful death.
-
1989: USDA Promulgated Parts 1 & 2 (Regulations) Implementing 1985 AWA
AmendmentsNonhuman Animal Research
In 1989, the United States Department of Agriculture (USDA) promulgated Parts 1 and 2 of the Animal Welfare Act (AWA) regulations, implementing key provisions of the 1985 AWA Amendments. These regulations established licensing and registration requirements for research facilities, dealers, exhibitors, and other entities covered by the AWA.
-
1990: USDA Promulgated Subpart B (Registration), Subpart C (Research
Facilities)Nonhuman Animal Research
In 1990, the United States Department of Agriculture (USDA) promulgated Subparts B and C as part of the regulations governing the Animal Welfare Act (AWA). Subpart B focused on the registration of research facilities that use non-human animals for experimentation, teaching, or testing purposes, requiring them to provide detailed information about their animal care and use programs. Subpart C outlined specific standards and requirements for research facilities to ensure the well-being and humane treatment of non-human animals under their care, covering aspects such as housing, veterinary care, and environmental enrichment.
-
1990: OPRR established Animal Welfare Division
Nonhuman Animal Research
In 1990, the Office for Protection from Research Risks (OPRR) established the Animal Welfare Division to oversee and enforce compliance with animal welfare regulations in research settings. Tasked with ensuring the humane care and use of non-human animals in projects funded by the Public Health Service (PHS), the Animal Welfare Division played a critical role in promoting ethical conduct and regulatory compliance among research institutions.
-
1991: USDA promulgated Part 3 (Standards), Amended Part 2 Subpart A
(Licensing) & Subpart D (AV & Adequate Vet Care)
Nonhuman Animal Research
In 1991, the United States Department of Agriculture (USDA) promulgated Part 3 of the Animal Welfare Act (AWA) regulations, establishing comprehensive standards for the humane care and treatment of non-human animals in research, exhibition, and commercial settings. These standards covered various aspects of animal care, including housing, feeding, veterinary care, and environmental enrichment, with the aim of ensuring the well-being and welfare of non-human animals under human care.
-
1992: Animal Enterprise Protection Act Passed By Congress
Nonhuman Animal Research
In 1992, the United States Congress passed the Animal Enterprise Protection Act (AEPA), legislation aimed at preventing and punishing acts of animal rights extremism and terrorism targeting animal enterprises, including research institutions, farms, and businesses involved in animal-related industries. The AEPA provided law enforcement agencies with expanded authority to investigate and prosecute individuals or groups engaged in activities such as vandalism, harassment, or intimidation against animal facilities or personnel.
-
1996: Guide Revision 7th edition
Nonhuman Animal Research
In 1996, the seventh edition of the Guide for the Care and Use of Laboratory Animals was published, providing updated guidelines and recommendations for the ethical and humane treatment of non-human animals in research, testing, and teaching environments. Developed by the Institute for Laboratory Animal Research (ILAR) and endorsed by the National Research Council (NRC), the Guide served as a foundational resource for researchers, veterinarians, and institutional animal care and use committees (IACUCs) responsible for overseeing animal welfare.
-
1993: FDA Guidance Issued - Study and Evaluation of Gender Differences in the
Clinical Evaluation of DrugsHuman Research
The NIH Revitalization Act of 1993, PL 103-43. The Food and
Drug Administration (FDA) Guidance and National Institutes of Health (NIH)
Policy, both released in 1993, sought to rectify the exclusion of women from
clinical research.
-
1995: Radiation ExperimentsHuman Research
-
1996: University of Rochester Bronchoscopy Case
Human Research
-
1997: NASA Principles for the Ethical Care and Use of Animals
Nonhuman Animal Research
In 1997, the National Aeronautics and Space Administration (NASA) established Principles for the Ethical Care and Use of Animals, outlining ethical guidelines and responsibilities for the use of non-human animals in NASA-funded research and spaceflight missions. These principles emphasized the importance of minimizing animal pain and distress, providing appropriate veterinary care, and ensuring the humane treatment of non-human animals throughout their involvement in research activities.
-
1998-2001: Research SuspendedHuman Research
OPRR, on behalf of the federal government, suspended federally-funded human
subjects research at 15 institutions due to various lapses in human subjects
research oversight. In many cases, the violations involved failure to obtain
adequate informed consent from research subjects.
-
1999: Jesse GelsingerHuman Research
-
2000: ICVAMNonhuman Animal Research
In 2000, the Interagency Coordinating Committee on the Validation of Alternative Methods (ICVAM) was established to coordinate federal efforts in the development, validation, and acceptance of alternative methods to animal testing. Comprising representatives from multiple government agencies, including the National Institutes of Health (NIH), the Food and Drug Administration (FDA), and the Environmental Protection Agency (EPA), ICVAM aimed to advance the use of scientifically valid alternative methods that reduce, refine, or replace the use of non-human animals in toxicological and biomedical research.
-
2000: OLAW EstablishedNonhuman Animal Research
In 2000, the Office of Laboratory Animal Welfare (OLAW) was established within the National Institutes of Health (NIH) to oversee and monitor compliance with the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals. Tasked with ensuring the ethical treatment of non-human animals in research funded by the PHS, OLAW provided guidance, oversight, and educational resources to research institutions and their Institutional Animal Care and Use Committees (IACUCs).
-
2000: OHRP EstablishedHuman Research
The Office for Human Research Protections (OHRP) was established in the
Office of the Assistant Secretary for Health to elevate its stature and
effectiveness. OHRP replaced the Office for Protection from Research Risks
(OPRR).
-
2000: NHRPAC EstablishedHuman Research
The National Human Research Protections Advisory Committee (NHRPAC) was
established to advise DHHS on human subject protection. The NHRPAC was later
replaced by SACHRP.
-
2000: Report of AVMA Panel on Euthanasia
Nonhuman Animal Research
In 2000, the American Veterinary Medical Association (AVMA) released a comprehensive report on euthanasia methods for non-human animals, providing guidance on humane euthanasia practices across various species and settings. Developed by a panel of veterinary experts, the report outlined recommended techniques, considerations, and criteria for selecting euthanasia methods based on factors such as species, size, and intended use.
-
2001: Ellen RocheHuman Research
Ellen Roche was a healthy 24-year-old lab technician at the Johns Hopkins
Asthma Center. She volunteered to take part in an experiment to understand
the natural defenses of healthy people against asthma. Roche was part of a
group that inhaled hexamethonium, a drug that induced a mild asthma attack.
Within 24 hours of inhaling the drug, Roche had lost one-third of her lung
capacity; she died within a month. The consent form she signed warned of
coughing, dizziness, and tightness in the chest, but not death. It called
hexamethonium a "medication" although its approval by the Food and Drug
Administration (FDA) (as a treatment for high blood pressure) had been
withdrawn in 1972.
-
2002: AWA Animal Definition Modified to Exclude Rats, Mice, Birds
Nonhuman Animal Research
In 2002, the definition of non-human animals under the Animal Welfare Act (AWA) was modified to exclude certain categories of non-human animals, specifically rats, mice, and birds bred for research purposes. This modification resulted in these non-human animals being excluded from the regulatory protections afforded to other covered species under the AWA.
-
2002: PHS Policy Modified JITNonhuman Animal Research
In 2002, the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals was modified to include Just-in-Time (JIT) procedures for the submission of Institutional Animal Care and Use Committee (IACUC) approvals. This modification streamlined the process for grant applicants by allowing them to submit IACUC approvals after peer review but before award issuance, reducing administrative burden and expediting the funding process for animal research projects.
-
2002: MOU NIH VANonhuman Animal Research
In 2002, a Memorandum of Understanding (MOU) was established between the National Institutes of Health (NIH) and the Department of Veterans Affairs (VA) to facilitate collaboration and coordination in animal research activities. The MOU outlined areas of mutual interest, such as the exchange of scientific information, resources, and expertise related to biomedical research involving non-human animals.
-
2002: HIPAA Privacy RuleHuman Research
The HIPAA Privacy Rule, originally issued in 2000, was modified in 2002.
The Privacy Rule addresses uses and disclosures of private health
information (PHI) for research purposes.
-
2003: SACHRPHuman Research
The Secretary's Advisory Committee on Human Research Protection (SACHRP)
replaces the NHRPAC. SACHRP is charged with providing advice to the DHHS on
matters relating to the responsible conduct of research involving human
subjects. This includes research issues involving special populations, such
as children, neonates, prisoners and the decisionally impaired; pregnant
women, embryos and fetuses; individuals and populations in international
studies; populations in which there are individually identifiable samples,
data or information; and investigator conflicts of interest.
-
2005-2009: SUPPORTHuman Research
The Surfactant, Positive Pressure, and Oxygenation Randomized Trial
(SUPPORT) was an early example of comparative effectiveness research (CER)
to determine the optimum oxygen therapy protocol for premature infants. The
National Institutes of Health (NIH)-sponsored multicenter trial randomly
assigned approximately 1,300 premature infants to either a low or high
oxygen treatment protocols. Considerable controversy about this study has
been raised, focusing on several issues. OHRP found that the parents in the
study were insufficiently informed of the risks involved, a conclusion that
has been disputed by some in the medical research community. Other
controversial issues include the random assignment of infants to treatment
groups, rather than assignment by a physician based on the infants' best
interests; and whether there was already sufficient medical knowledge at the
time, making a randomized trial inappropriate.
-
2006: Animal Enterprise Terrorism Act Passed By Congress
Nonhuman Animal Research
In 2006, the Animal Enterprise Terrorism Act (AETA) was passed by the United States Congress, aiming to address acts of terrorism and violence targeting animal enterprises, including research institutions, agricultural facilities, and businesses involved in animal-related industries. The AETA expanded federal law enforcement's authority to investigate and prosecute individuals engaged in activities that cause economic damage, disruption, or fear to animal enterprises through illegal acts such as vandalism, harassment, or intimidation.
-
2007: AVMA Guidelines On Euthanasia
Nonhuman Animal Research
In 2007, the American Veterinary Medical Association (AVMA) released updated guidelines on euthanasia methods for non-human animals, providing evidence-based recommendations and best practices to ensure the humane and ethical euthanasia of non-human animals across various species and settings. The guidelines addressed considerations such as the selection of appropriate euthanasia agents, techniques, and procedures based on species-specific factors, as well as the importance of trained personnel and compassionate care during the euthanasia process.
-
2010: Guide Revision 8th Edition
Nonhuman Animal Research
In 2010, the Guide for the Care and Use of Laboratory Animals, published by the National Research Council (NRC), underwent its eighth edition revision, providing updated standards and recommendations for the housing, care, and use of non-human animals in research, testing, and teaching environments. The revised edition incorporated new scientific knowledge, advancements in animal welfare practices, and changes in regulatory requirements to ensure the continued improvement of animal care and research standards.
-
2010: Havasupai SettlementHuman Research
The Havasupai tribe settled with Arizona State University (ASU) concerning
their claim that blood collected in the 1990s by ASU researchers to study
the genetic origins of diabetes among tribal members was used for unrelated
projects including studies of schizophrenia, inbreeding, and theories of the
tribe's geographical origins that contradict their traditional stories. The
Havasupai had consented to the diabetes study, but the other studies were
performed without their knowledge or consent.
-
2010: Guatemala Syphilis StudyHuman Research
In 2010, Dr. Susan Reverby, a medical historian, announced her discovery of
a syphilis study conducted in Guatemala from 1946 to 1948. In the study,
funded by the U.S. government, nearly 700 Guatemalans — prison inmates,
mental patients and soldiers — were deliberately infected with venereal
diseases in what was meant as an effort to test the effectiveness of
penicillin. Representatives of the U.S. government apologized to the
government of Guatemala and the survivors and descendants of those infected,
calling the experiments "clearly unethical."
-
2010: The Immortal Life of Henrietta Lacks
Human Research
-
2011-2018: Process to Revise the Common Rule
Human Research
-
2014: Ebola Outbreak in West Africa
Human Research
-
2014: Facebook Emotional Contagion Study
Human Research
The social media platform Facebook conducted a massive experiment in which
the news feeds of over 600,000 Facebook users were manipulated. The
experiment found that emotional contagion, a well-characterized phenomenon
in offline environments, also occurs online. The study, published by the
Proceedings of the National Academy of Sciences in 2014, raised widespread
ethical concerns around the ideas of privacy, industry-academic
partnerships, and the nature of informed consent.
-
2016: 21st Century Cures ActHuman Research
The 21st Century Cures Act was signed into law, providing broad support for
biomedical research and implementing measures to reduce administrative
burden. Impacts to human research protections included allowances for waiver
or alteration of informed consent for Food and Drug Administration
(FDA)-regulated minimal risk studies, and for use of a single IRB for
multisite medical device studies, as well as a call for harmonization of FDA
regulations and the Common Rule. Provisions to support increased diversity
among clinical research subjects, protect vulnerable populations in
research, and enhance privacy protections for subjects' sensitive data, were
also included.
-
2018-2020: Single IRB MandatesHuman Research
The National Institutes of Health (NIH) mandated that all multi-site
cooperative research funded by NIH must rely upon ethical review and
approval conducted by a single IRB of record. The revised Common Rule
followed suit and included a requirement for single IRB review of
cooperative research to take effect in 2020.
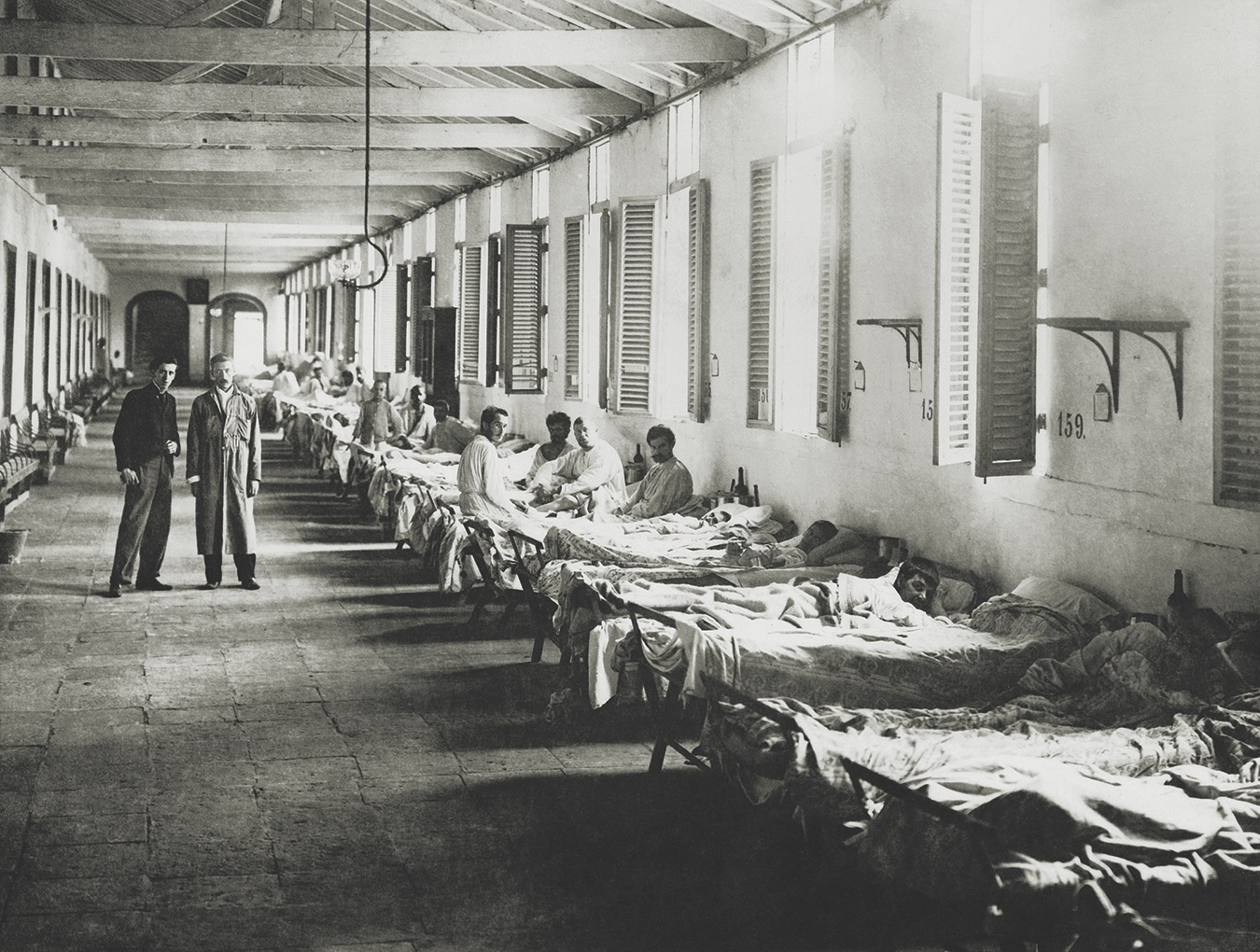 Walter Reed was a Major in the U.S. Army appointed to study tropical
diseases in Cuba. His research on yellow fever revealed that the disease is
transmitted through mosquitoes biting humans rather than from person to
person.
Walter Reed was a Major in the U.S. Army appointed to study tropical
diseases in Cuba. His research on yellow fever revealed that the disease is
transmitted through mosquitoes biting humans rather than from person to
person.  In 1930, the Hygienic Laboratory was re-designated as the National Institute of Health (NIH). The establishment NIH marked a pivotal moment in medical research and public health. The NIH became a central hub for coordinating biomedical research efforts, laying the groundwork for numerous groundbreaking discoveries and advancements in healthcare.
In 1930, the Hygienic Laboratory was re-designated as the National Institute of Health (NIH). The establishment NIH marked a pivotal moment in medical research and public health. The NIH became a central hub for coordinating biomedical research efforts, laying the groundwork for numerous groundbreaking discoveries and advancements in healthcare.
 The US Public Health Service (PHS) Tuskegee Study of Untreated Syphilis in
the Negro Male is sometimes called the "Tuskegee Study" because it took
place in Tuskegee, Alabama. The press exposure of this study set the stage
for the National Research Act, which requires IRB review of all
federally-funded studies.
The US Public Health Service (PHS) Tuskegee Study of Untreated Syphilis in
the Negro Male is sometimes called the "Tuskegee Study" because it took
place in Tuskegee, Alabama. The press exposure of this study set the stage
for the National Research Act, which requires IRB review of all
federally-funded studies.  The Nazi Doctors' Trial began in 1946 following World War II. Criminal
proceedings were brought against 23 Germans for participating in war crimes
and crimes against humanity. Sixteen were found guilty and seven were
sentenced to death because of their participation in experiments on human
beings that involved painful and frequently fatal procedures.
The Nazi Doctors' Trial began in 1946 following World War II. Criminal
proceedings were brought against 23 Germans for participating in war crimes
and crimes against humanity. Sixteen were found guilty and seven were
sentenced to death because of their participation in experiments on human
beings that involved painful and frequently fatal procedures.  Researchers at the Willowbrook State School in New York, an institution
for mentally disabled children, deliberately infected children with a mild
form of hepatitis during a study of the progression, prevention and
treatment of viral hepatitis that extended from 1956 to 1972. The results
eventually contributed to the development of a successful hepatitis vaccine
but were controversial since the study involved healthy, institutionalized
children and parental consent issues.
Researchers at the Willowbrook State School in New York, an institution
for mentally disabled children, deliberately infected children with a mild
form of hepatitis during a study of the progression, prevention and
treatment of viral hepatitis that extended from 1956 to 1972. The results
eventually contributed to the development of a successful hepatitis vaccine
but were controversial since the study involved healthy, institutionalized
children and parental consent issues.  In the late 1950s, Thalidomide was approved as a sedative in Europe but
the Food and Drug Administration (FDA) had not approved it for use in the
U.S. At that time, the FDA had only a limited role approving drugs in the
U.S., which allowed the manufacturer to give samples to U.S. physicians and
pay them to study its safety and efficacy.
In the late 1950s, Thalidomide was approved as a sedative in Europe but
the Food and Drug Administration (FDA) had not approved it for use in the
U.S. At that time, the FDA had only a limited role approving drugs in the
U.S., which allowed the manufacturer to give samples to U.S. physicians and
pay them to study its safety and efficacy.  This social science study was conducted in an attempt to understand – from
a psychological standpoint – the role of obedience to authority: why
ordinary people behave in ways that seem inhumane outside a certain context.
The research design was to have an authority figure instruct subjects to do
something that they would not do under ordinary circumstances.
This social science study was conducted in an attempt to understand – from
a psychological standpoint – the role of obedience to authority: why
ordinary people behave in ways that seem inhumane outside a certain context.
The research design was to have an authority figure instruct subjects to do
something that they would not do under ordinary circumstances.  In 1971, researcher Philip Zimbardo conducted a study of the psychology of
imprisonment by setting up a mock prison using volunteer college student
subjects to assume the roles of prisoners and guards. The student "guards"
brutalized the student "prisoners."
In 1971, researcher Philip Zimbardo conducted a study of the psychology of
imprisonment by setting up a mock prison using volunteer college student
subjects to assume the roles of prisoners and guards. The student "guards"
brutalized the student "prisoners."  A healthy 19-year-old college student died after volunteering for a
medical research study in which University of Rochester researchers
performed a bronchoscopy under general anesthesia. An investigation revealed
that researchers exceeded the maximum dosage of lidocaine established by the
research protocol.
A healthy 19-year-old college student died after volunteering for a
medical research study in which University of Rochester researchers
performed a bronchoscopy under general anesthesia. An investigation revealed
that researchers exceeded the maximum dosage of lidocaine established by the
research protocol.
 Rebecca Skloot publishes The Immortal Life of Henrietta Lacks, a
book that describes the short life of Henrietta Lacks, who died in 1951 from
cervical cancer at age 31. Her cancer cells were the originators of the HeLa
cell line, an incredibly robust line that has contributed to countless
scientific advances over the years. Her descendants' realization of their
mother's 'immortality' (for which she received no compensation or
recognition) is the focus of the book. The book sparked a national dialogue
about the ethics of collection and subsequent use of biological tissue for
research purposes.
Rebecca Skloot publishes The Immortal Life of Henrietta Lacks, a
book that describes the short life of Henrietta Lacks, who died in 1951 from
cervical cancer at age 31. Her cancer cells were the originators of the HeLa
cell line, an incredibly robust line that has contributed to countless
scientific advances over the years. Her descendants' realization of their
mother's 'immortality' (for which she received no compensation or
recognition) is the focus of the book. The book sparked a national dialogue
about the ethics of collection and subsequent use of biological tissue for
research purposes.
 The outbreak of the Ebola virus in West Africa killed over 11,000 people
across six countries in just under two years. The urgent need to treat the
disease led to rapid approval of clinical trials in affected areas, raising
novel ethical questions about researching unproven therapies in public
health emergencies.
The outbreak of the Ebola virus in West Africa killed over 11,000 people
across six countries in just under two years. The urgent need to treat the
disease led to rapid approval of clinical trials in affected areas, raising
novel ethical questions about researching unproven therapies in public
health emergencies.